A Fatal Case of Rivaroxaban-Induced Hepatotoxicity in a Patient with Hepatocellular Carcinoma
Omar Alshuwaykh, Michael Krier
Med Sci Case Rep 2018; 5:33-35
DOI: 10.12659/MSCR.910939
Available online: 2018-05-14
Published: 2018-05-14
BACKGROUND:
Rivaroxaban has been associated with a small risk of hepatotoxicity. The 2014 report of the American College of Cardiology, American Heart Association Task Force on Practice Guidelines and the Heart Rhythm Society (AHA/ACC/HRS) recommended monitoring hepatic function prior to initiation of rivaroxaban and at least annually.
CASE REPORT:
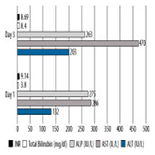
A 77-year-old white woman with a history of recently diagnosed hepatocellular carcinoma and bilateral pulmonary embolism was admitted to the hospital with complicated UTI. The patient was recently started on rivaroxaban 1 month prior to admission. Her liver function tests were within normal limits at that time. During this admission, her ALT, AST, and ALP were 2.2, 7.7, and 2.4 times the upper limit of normal range (ULN), respectively. Total bilirubin was 3.8 mg/dl and INR was 9.14. Serologic and virologic testes for acute and chronic viral hepatitis, autoimmune hepatitis, CMV, EBV, and HSV were all negative. Biliary obstruction, primary biliary cirrhosis, sclerosing cholangitis, hemochromatosis, and Wilson disease were all ruled out. Rivaroxaban was discontinued upon admission. Liver enzymes continued to trend up on the third day to ALT and AST of 3.4 and 12.7 times ULN, respectively. ALP trended down to 2.3 times ULN. Total bilirubin was 8.4 mg/dl and INR was 8.69. Her RUCAM score was 6, which suggested that rivaroxaban-induced hepatotoxicity was probable. The patient then developed severe hepatic encephalopathy and her family elected to change her status to comfort measures only, after which the patient died from liver failure.
CONCLUSIONS:
Physicians need to be more attentive in monitoring liver function tests regularly when starting rivaroxaban, especially in patients with pre-existing liver diseases like hepatocellular carcinoma.
Keywords: hepatocellular carcinoma, Factor Xa Inhibitors, Drug Induced Liver Injury, Rivaroxaban





